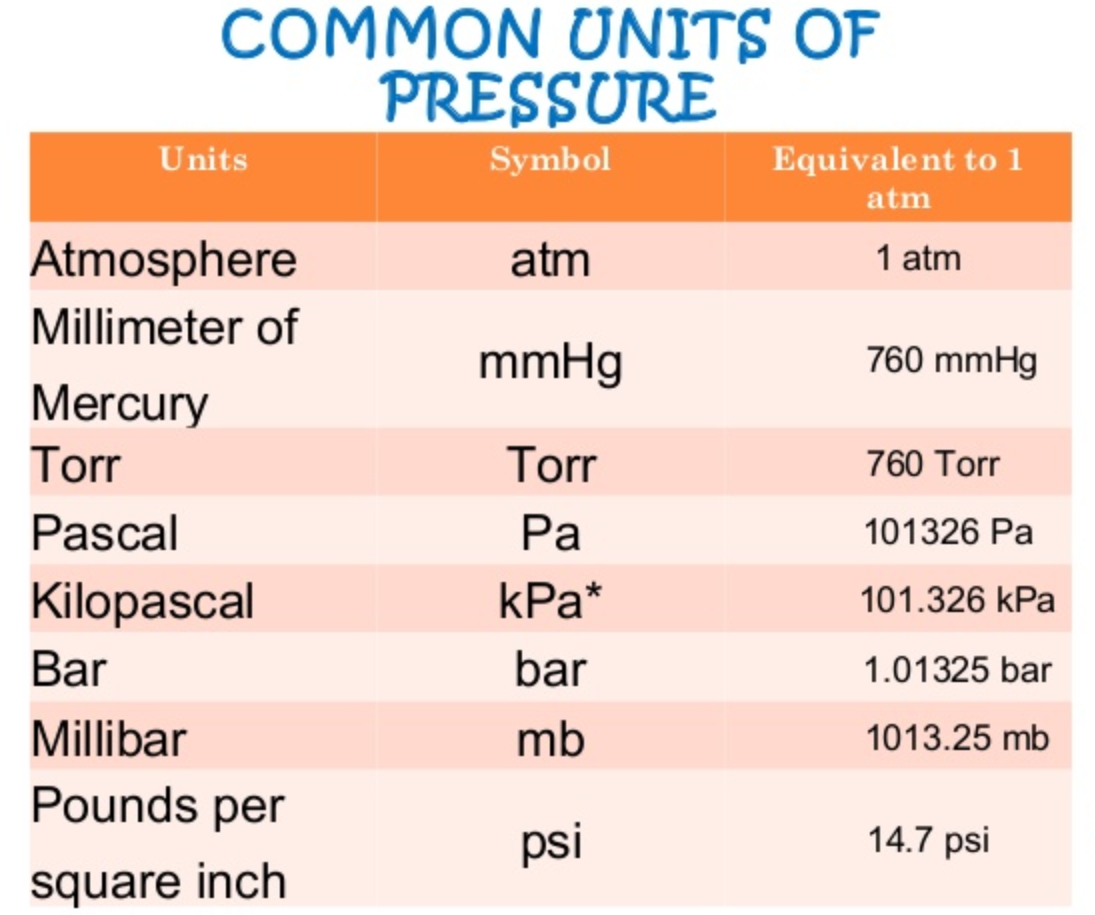
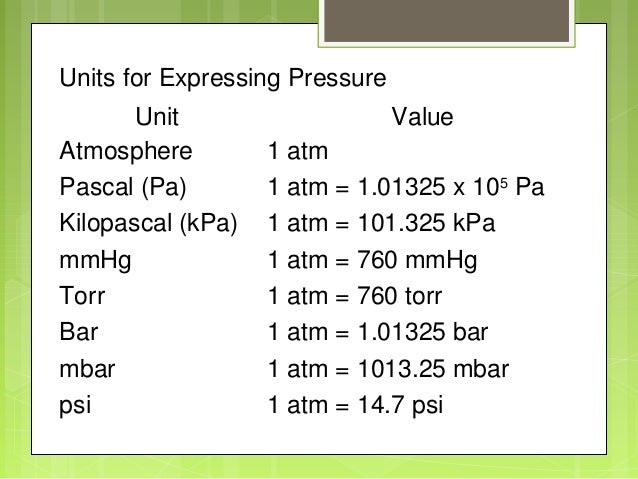
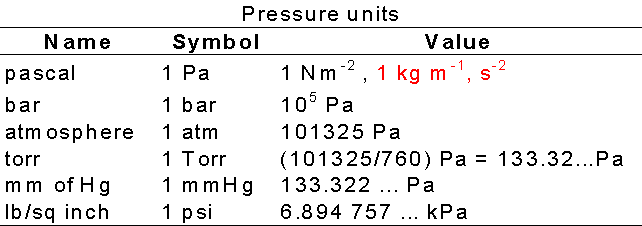
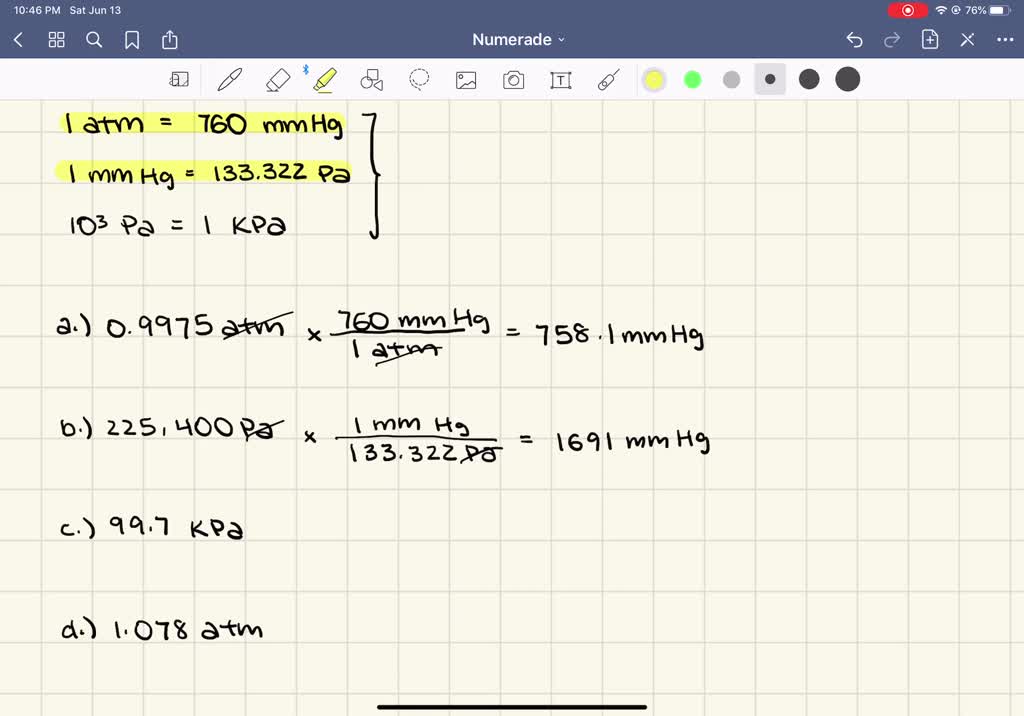
1 atm = 760 mmHg = 760 torr = 1.01325 x 10 Pa = 14.7 lb/in R = 0.0821 L atm mol K or 8.314 J mol K 1 J = 1 Kg m s2 -2 1 Pa = 1

Gas Pressure Unit Conversions - torr to atm, psi to atm, atm to mm Hg, kpa to mm Hg, psi to torr - YouTube

Pressure 1 atmosphere ~ 1 bar ~ 760 mm Hg ~ 760 torr ~ 100,000 Pa Ion gauges read in mbar i.e. 1x mbar = 1x atm. Sometimes ion gauges read. - ppt download
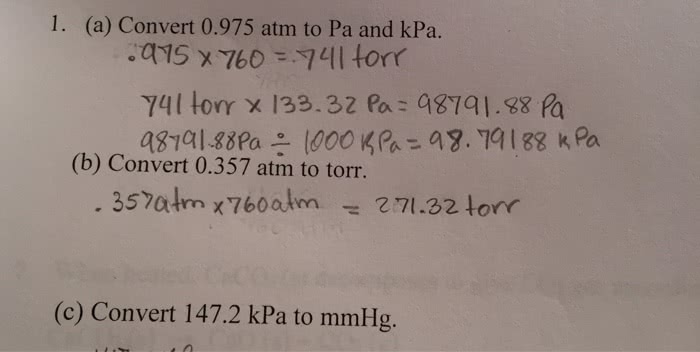
OneClass: 1. (a) Convert 0.975 atm to Pa and kPa. .qy5 x 760 = 41 torr (b) Convert 0.357 atm to torr....

SOLVED: Convert 661,000 pascal, pa into torr: 1atm = 760 torr = 760 mmHg = 101,325 pa 101.325 kpa 1.01325 barr 14.696 p.s.i. 29.921 inHg (inHg and p.s.i. are approximations of the metric pressures and are subject to significant figures)

SOLVED:Make the indicated pressure conversions. a. 1.54 ×10^5 Pa to atmospheres b. 1.21 atm to pascals c. 97,345 Pa to mm Hg d. 1.32 kPa to pascals


![❓ Need to convert mm of mercury to kilo [hecto] pascal ❓ ❓ Need to convert mm of mercury to kilo [hecto] pascal ❓](http://www.eng.inshanisha.com/wp-content/uploads/2021/08/750-mmhg-1024x576.png)