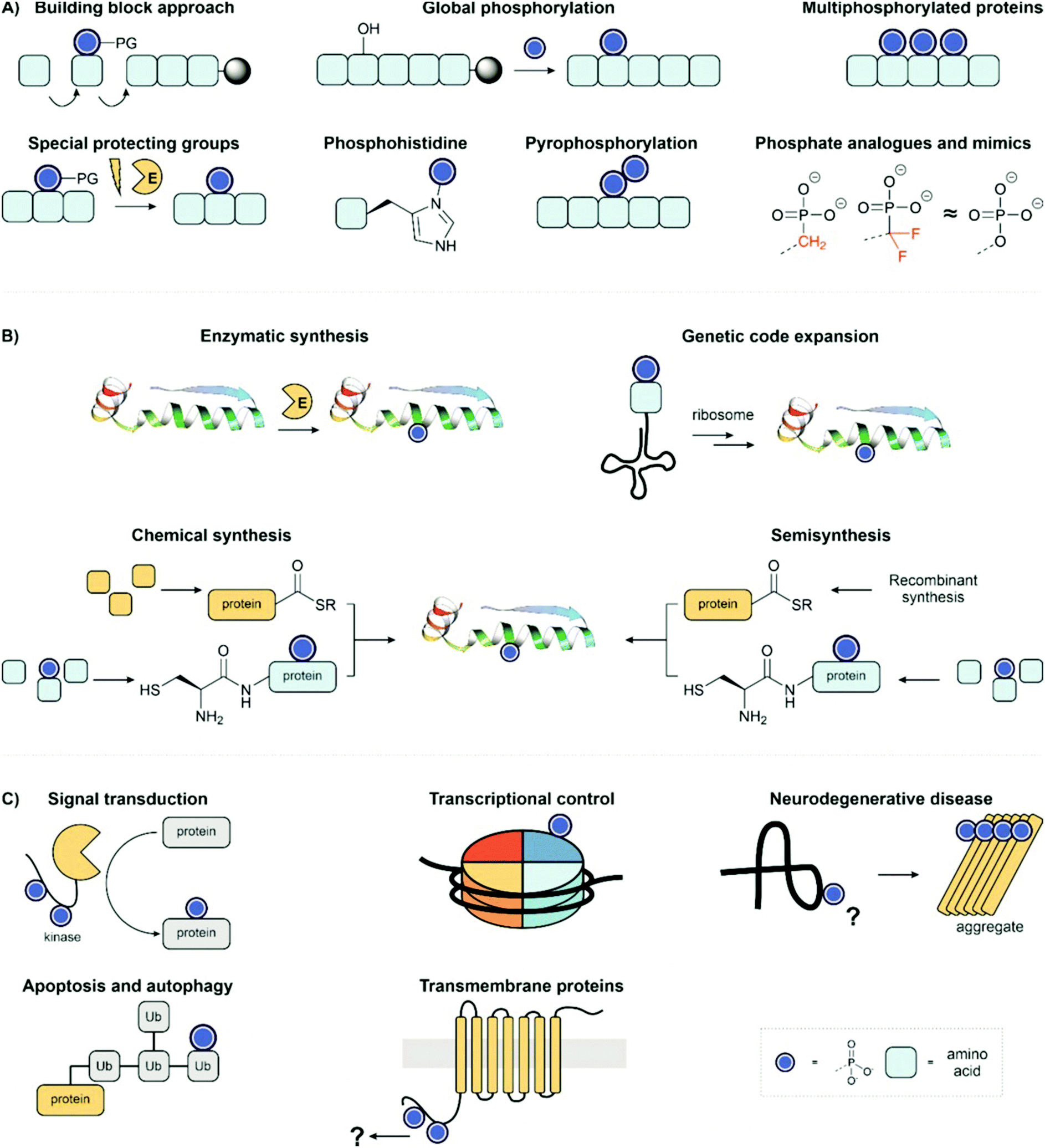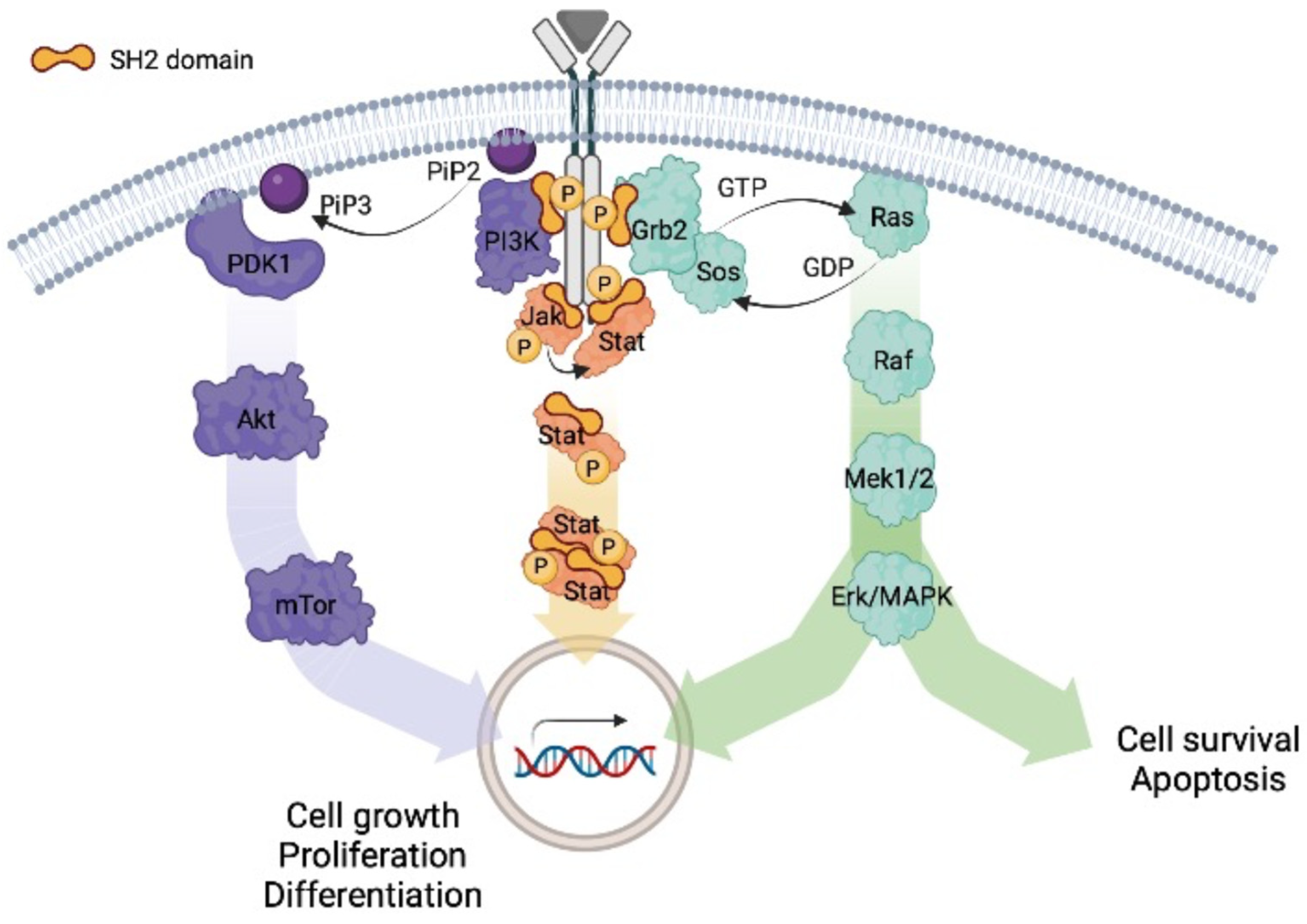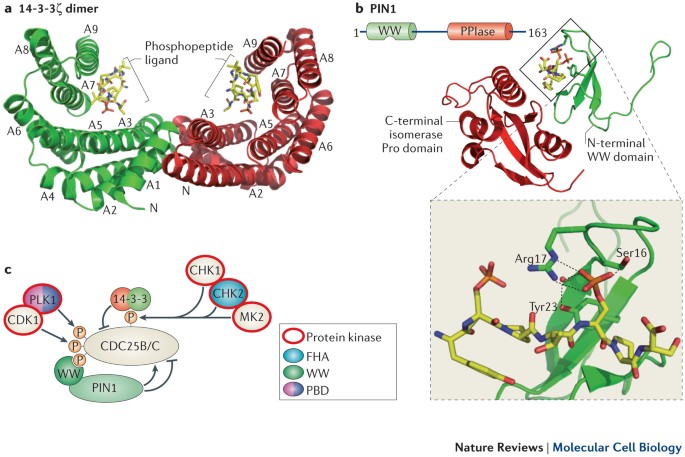
Phospho-Ser/Thr-binding domains: navigating the cell cycle and DNA damage response | Nature Reviews Molecular Cell Biology

Phospho-Ser/Thr-binding domains: navigating the cell cycle and DNA damage response | Nature Reviews Molecular Cell Biology

RNA Polymerase II C-Terminal Domain: Tethering Transcription to Transcript and Template | Chemical Reviews

PDF) Selectivity and promiscuity in the interaction network mediated by protein recognition modules | Luisa Castagnoli and Serena Paoluzi - Academia.edu

Effective Disruption of Phosphoprotein−Protein Surface Interaction Using Zn(II) Dipicolylamine-Based Artificial Receptors via Two-Point Interaction | Journal of the American Chemical Society

PDF) Structural basis for the interaction of the free SH2 domain EAT-2 with SLAM receptors in hematopoietic cells

Dual recognition of phosphoserine and phosphotyrosine in histone variant H2A.X by DNA damage response protein MCPH1. - Abstract - Europe PMC

Uncovering the Mechanism of Forkhead-Associated Domain-Mediated TIFA Oligomerization That Plays a Central Role in Immune Responses | Biochemistry

A Methodological Assessment and Characterization of Genetically-Driven Variation in Three Human Phosphoproteomes | Scientific Reports

Kinase inhibition profiles as a tool to identify kinases for specific phosphorylation sites | Nature Communications

Conserved interdomain linker promotes phase separation of the multivalent adaptor protein Nck | PNAS
Semi-Supervised Prediction of SH2-Peptide Interactions from Imbalanced High-Throughput Data | PLOS ONE