
Site-selective amination and/or nitrilation via metal-free C(sp 2 )–C(sp 3 ) cleavage of benzylic and allylic alcohols - Chemical Science (RSC Publishing) DOI:10.1039/D2SC00758D

Cu(OTf)2-catalyzed Beckmann Rearrangement of Ketones Using Hydroxylamine-O-sulfonic Acid (HOSA). - Abstract - Europe PMC
Zinc(II)-Catalyzed Synthesis of Secondary Amides from Ketones via Beckmann Rearrangement Using Hydroxylamine-O-sulfonic Acid in

The Mechanochemical Beckmann Rearrangement: An Eco-efficient “Cut-and-Paste” Strategy to Design the “Good Old Amide Bond” | ACS Sustainable Chemistry & Engineering

Zinc(II)-Catalyzed Synthesis of Secondary Amides from Ketones via Beckmann Rearrangement Using Hydroxylamine-O-sulfonic Acid in Aqueous Media

Cu(OTf)2-catalyzed Beckmann Rearrangement of Ketones Using Hydroxylamine-O-sulfonic Acid (HOSA). - Abstract - Europe PMC
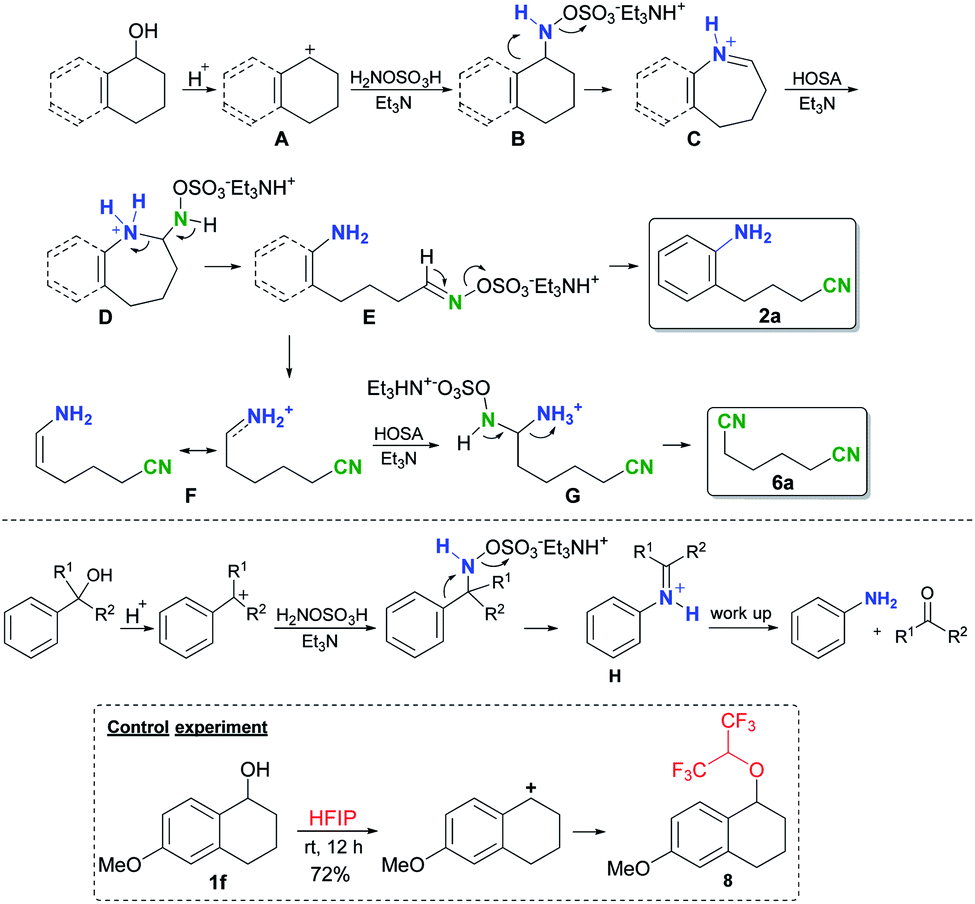
Site-selective amination and/or nitrilation via metal-free C(sp 2 )–C(sp 3 ) cleavage of benzylic and allylic alcohols - Chemical Science (RSC Publishing) DOI:10.1039/D2SC00758D

The Mechanochemical Beckmann Rearrangement: An Eco-efficient “Cut-and-Paste” Strategy to Design the “Good Old Amide Bond” | ACS Sustainable Chemistry & Engineering
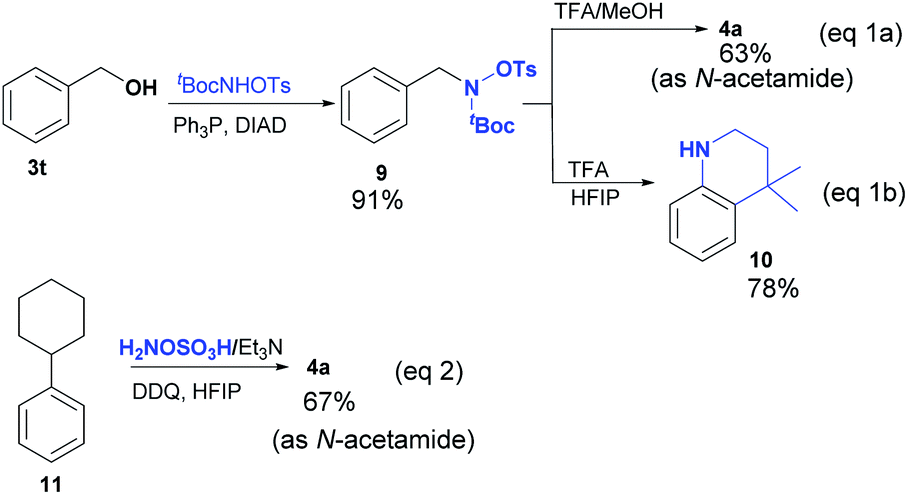
Site-selective amination and/or nitrilation via metal-free C(sp 2 )–C(sp 3 ) cleavage of benzylic and allylic alcohols - Chemical Science (RSC Publishing) DOI:10.1039/D2SC00758D















