Calculate the number of KJ of heat necessary to raise the temperature of 60.0g of aluminium from.... - YouTube

The specific heat of the PCM is 2.3 KJ/kg/°C, the density of the PCM is... | Download Scientific Diagram
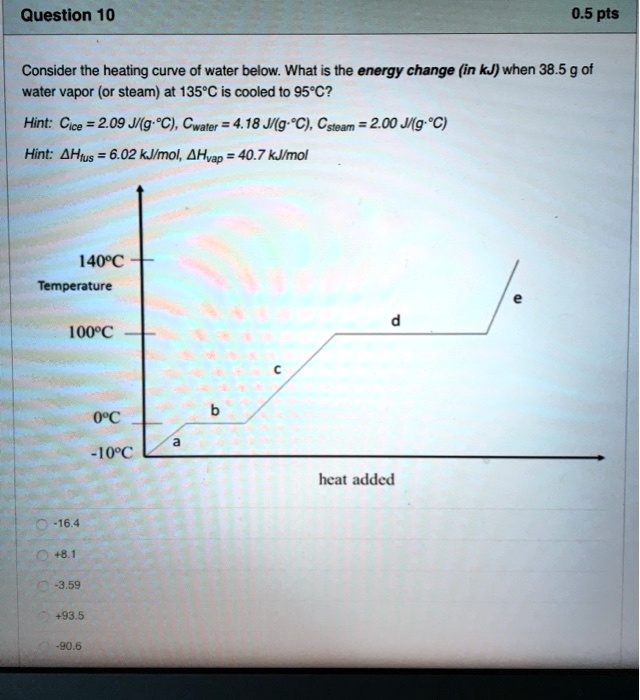
SOLVED: Question 10 0.5 pts Consider the heating curve of water below: What is the energy change (in kJ) when 38.5 9 ot water vapor (or steam) at 135*C is cooled to

The heat of combustion of C(graphite) is - 393.5kJ mol^-1 . The heat of formation of CO2 from graphite is kJ mol^-1 .

A heat pump is used to maintain a house at 25 ? C by extracting heat from the outside air on a day when the outside air temperature is 4 ? C.







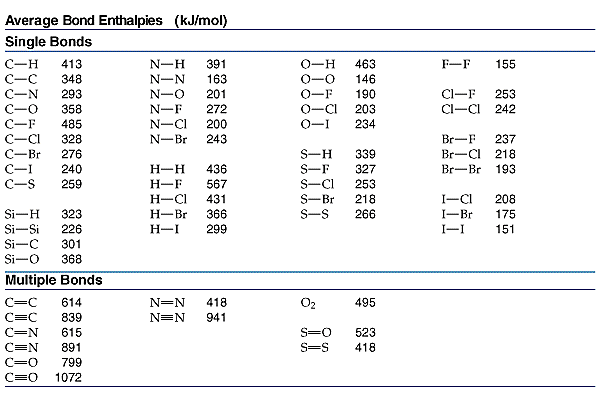

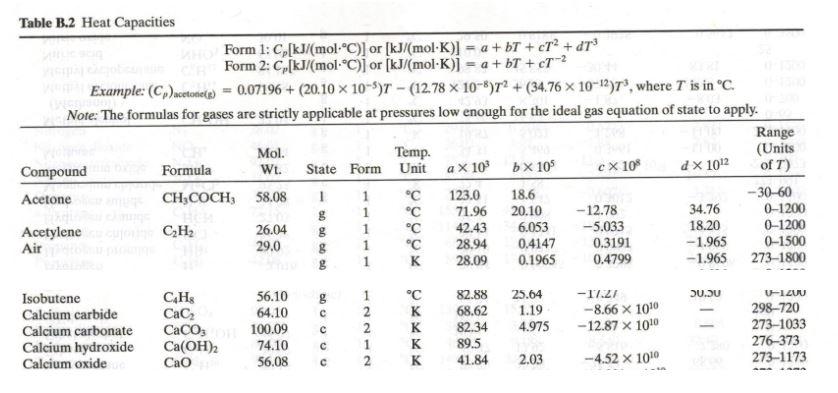
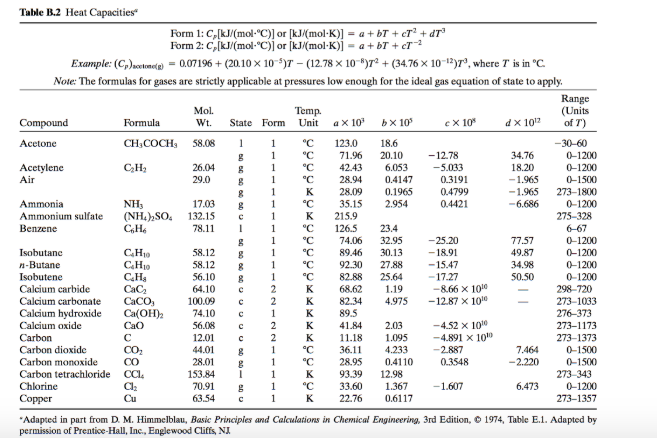
![Solved Table B.2 Heat Capacities Form 1: C1kJ (mol·°C)] or | Chegg.com Solved Table B.2 Heat Capacities Form 1: C1kJ (mol·°C)] or | Chegg.com](https://d2vlcm61l7u1fs.cloudfront.net/media%2Fd1e%2Fd1ec8c67-abd1-4bf2-88d0-ce524e9ef2b2%2FphpIIt2ym.png)