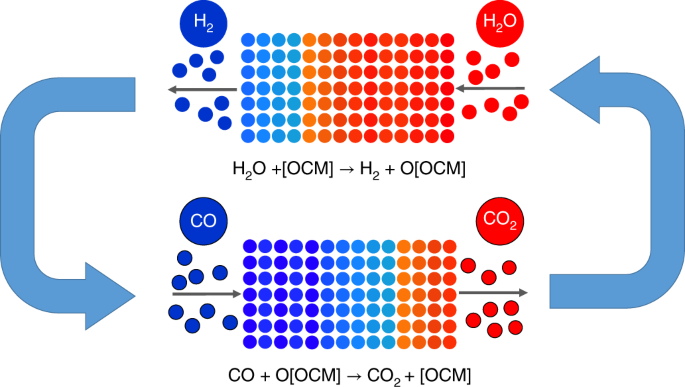
Overcoming chemical equilibrium limitations using a thermodynamically reversible chemical reactor | Nature Chemistry

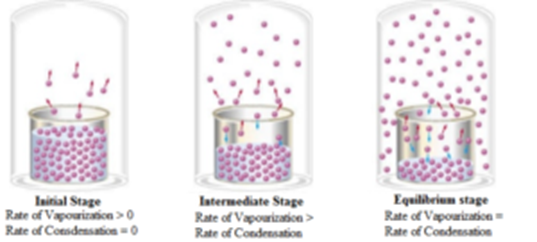
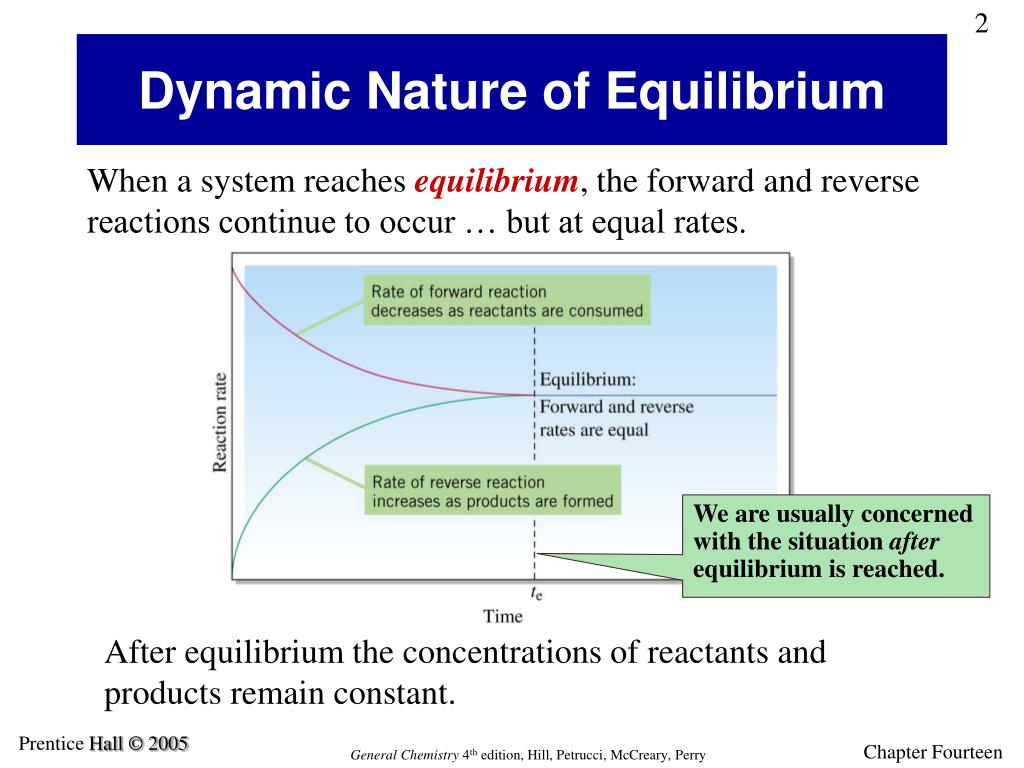
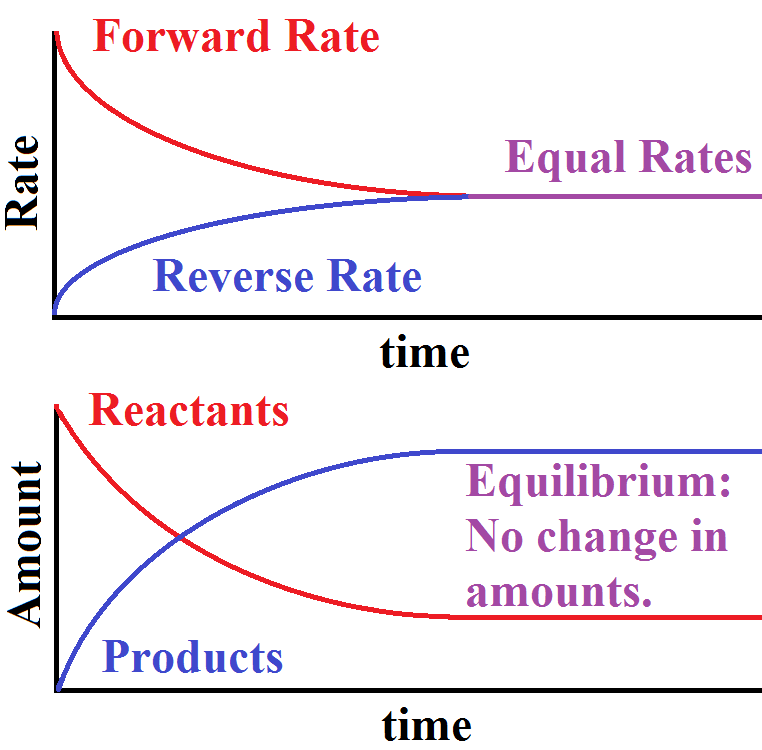
SOLVED: Chemical equilibrium is considered dynamic in nature because: the chemical reactions achicve equilibrium rapidly: both forward and backward reactions occur with the same rate. the concentration of reactants and products become

Chemistry 11Chapter 7 Equilibrium In Chemical Processes #Dynamic Nature Of Chemical Equilibrium - YouTube

Why chemical equilibrium is dynamic in nature - Chemistry - Equilibrium - 10747085 | Meritnation.com