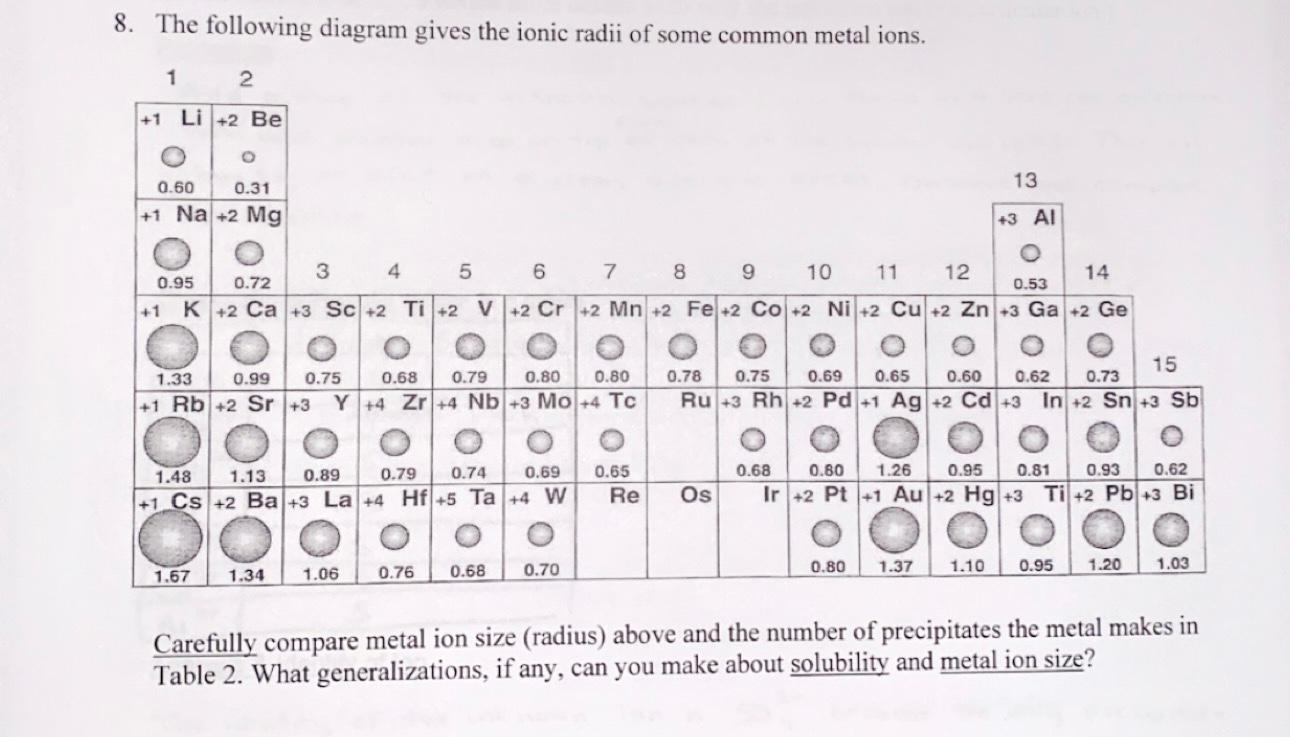
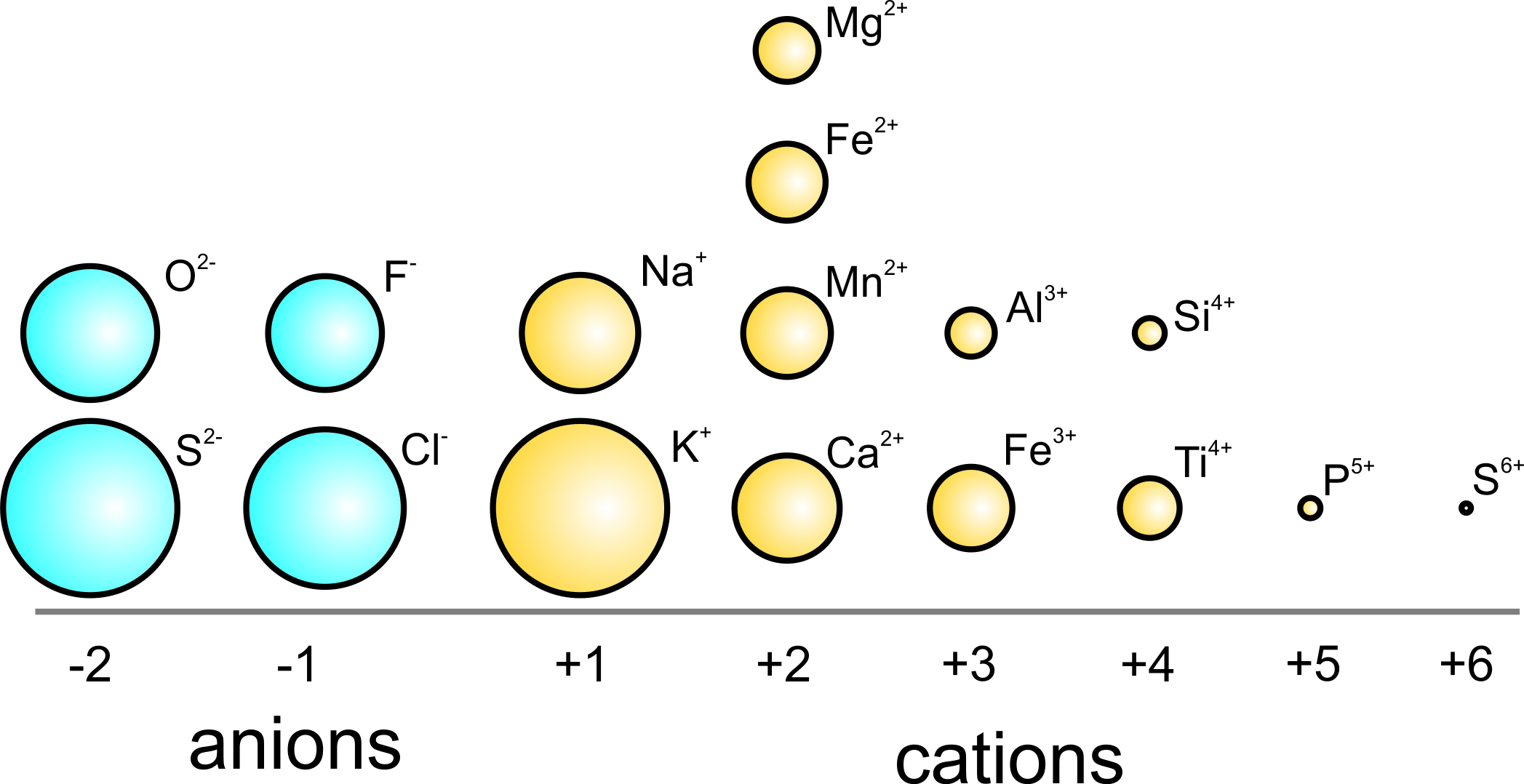
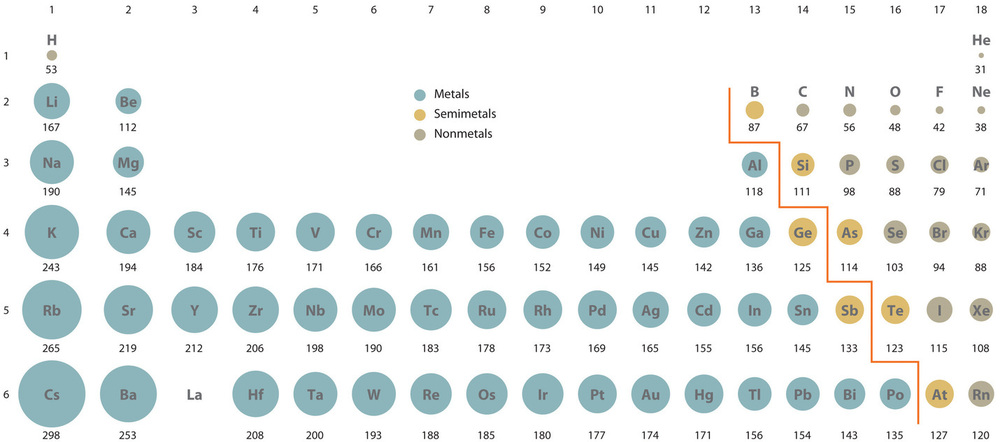
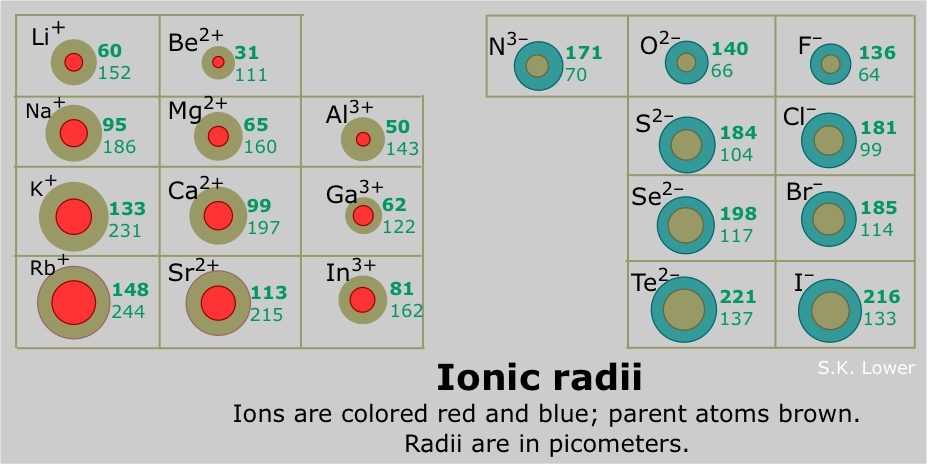
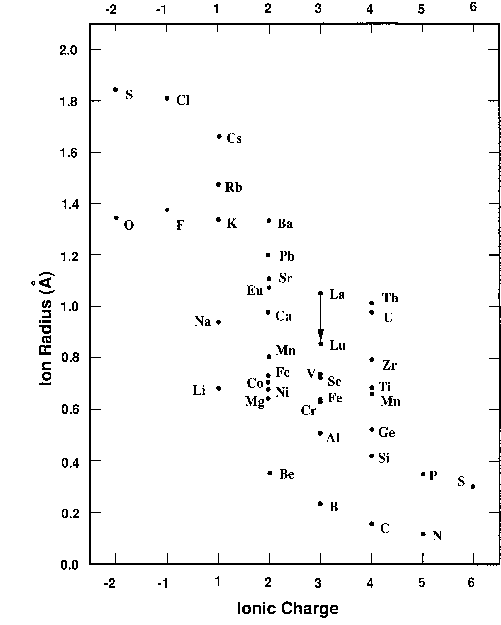
How to Use Coulomb's Law to Understand the Strength of Interactions between Cations & Anions Based on Ion Size | Chemistry | Study.com
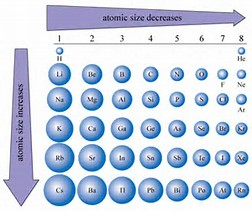
How does ionic size relate to the size of their particular parent atom? In your answer discuss the relative size of the ions of potassium, bromine, and selenium. | Socratic
![PDF] Relation between the ion size and pore size for an electric double-layer capacitor. | Semantic Scholar PDF] Relation between the ion size and pore size for an electric double-layer capacitor. | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/ecb3a41c65a11f0bec14f6fedbb9ac8b9903abbf/2-Figure3-1.png)
PDF] Relation between the ion size and pore size for an electric double-layer capacitor. | Semantic Scholar
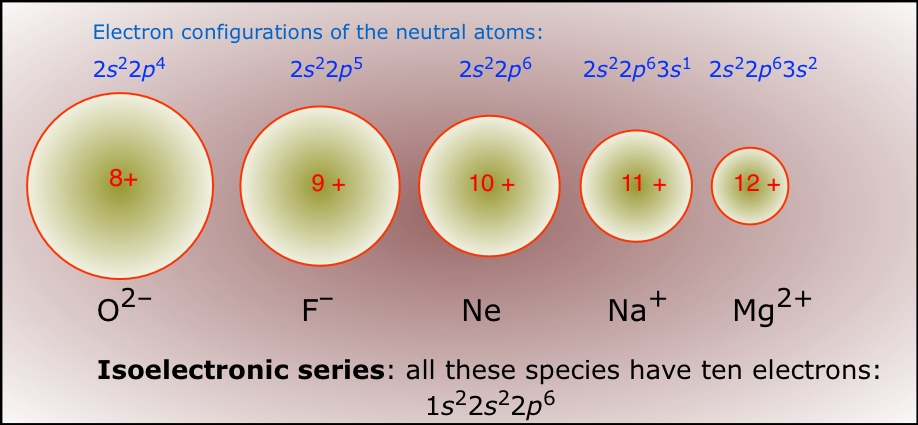
The primary reason sodium ions are smaller than sodium atoms is that the ion has only two shells of electrons (the atom has three). Some resources suggest the ion gets smaller since

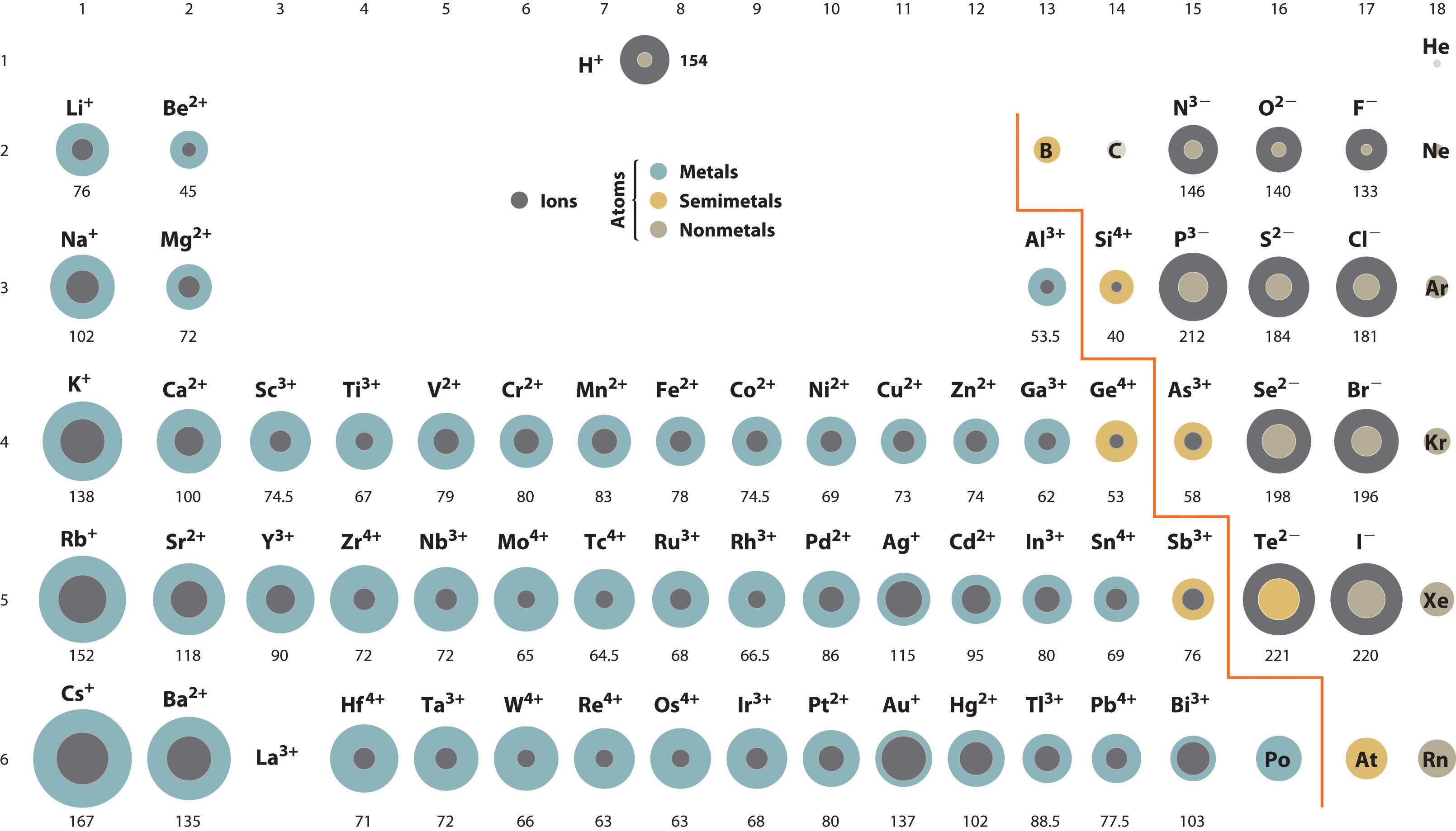
Periodic Trends in Ionic Size - Chemistry | Socratic | Ionic radius, Chemistry education, Teaching chemistry
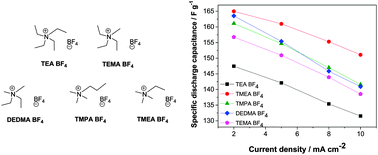
The effect of the ionic size of small quaternary ammonium BF4 salts on electrochemical double layer capacitors - Physical Chemistry Chemical Physics (RSC Publishing)