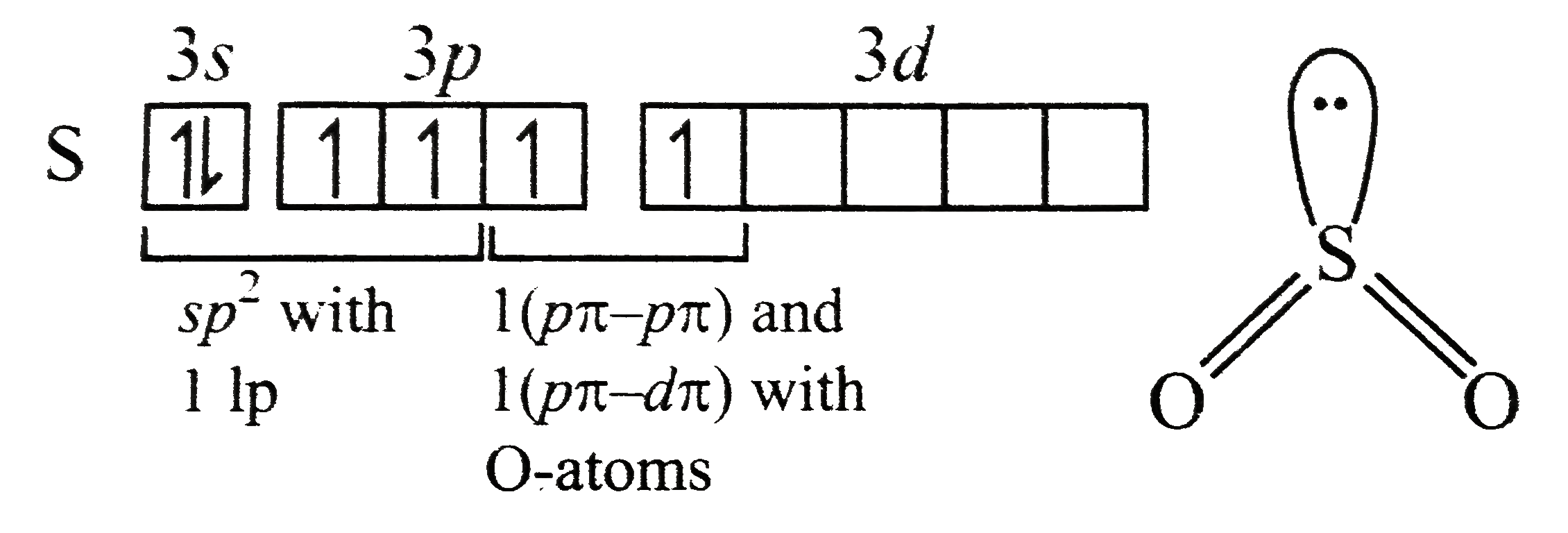The high reactivity of alkyl halides can be explained in terms of nature of C-X bond which is a highly polarised covalent bond. This polarity is responsible for the nucleophilic substitution reaction

Legendary Druid | The Fifth Legendary Class! Base Features for levels 1-30, infinite leveling beyond 30, and tons of customizability added to the base class! | Become a true guardian of nature!

Nature of bond can be predicted on the basis of electronativity of bonded atoms, greater difference in electronegativity (X), more will be the polarity of bond, and polar bond are easily broken